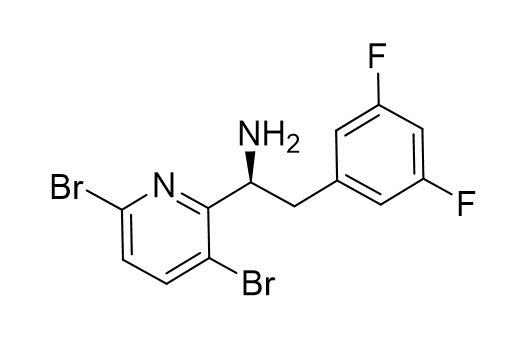
Synthesis of Fragment A of Lenacapavir
Medicines for All Institute (M4ALL) demonstrated a 7-step route to make Fragment A (Frag A) that offers an overall raw material cost (RMC) reduction of 33-41%.
Key Assumptions
- Projected material cost reductions are significantly impacted by market dynamics for starting materials and reagents, and economies of scale related thereto. Please reach out to M4ALL for more information.
- The baseline route refers to the Gilead’s route described in the process development report below.
Scientific Highlights
- We demonstrate a chiral resolution-based approach to produce Frag A from 3,6-dibromo- 2-methylpyridine.
- The key transformations include an intensified two-step aldehyde synthesis, a telescoped three-step racemic amine synthesis, and a Gilead-pioneered dynamic kinetic resolution (DKR) with N-acetyl-D-leucine (NADL).
- Process development of this 7-step strategy enables the production of Frag A as a single enantiomer in 25-30% overall isolated yield, demonstrated at scales up to 200 grams.
Summary of Process Development Work on the Synthesis of Frag A of Lenacapavir
PAT Use in the Dynamic Kinetic Resolution Reaction of Lenacapavir Fragment A and its Temperature Dependence
Medicines for All (M4ALL) utilized PAT to further understand and characterize the rate of a dynamic kinetic resolution (DKR) reaction used to produce Fragment A based on reaction temperature.
Scientific Highlights
- Inline FTIR was utilized for real time measurement with a complementary offline GC-MS method to track reaction progress.
- Reaction cycle times ranged between 20 hrs-98 hrs (0.8 days-4.1 days) depending on temperature (higher temperature = shorter cycle time).
- Isolated yields (33%-58%), ee (67%-100%) and impurity generation for (S)-A1.5-NADL were temperature dependent (higher temperature = lower yield/ee and new impurities).
GFN-002A Len Frag A DKR Summary Report