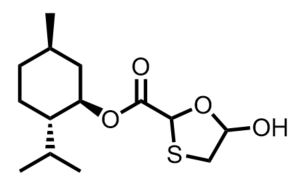
Supply Centered Synthesis to Current Late-State Intermediate: (1R,2S,5R)-2-isopropyl-5-methylcyclohexyl 5-hydroxy-1,3-oxathiolane-2-carboxylate
Intermediate
Summary
We report an efficient one-pot protocol for the preparation of (1R,2S,5R)-2-isopropyl-5-methylcyclohexyl 5-hydroxy-1,3-oxathiolane-2-carboxylate from low cost commodity feedstocks. Sulfenyl chloride chemistry is used to construct the framework for the oxathiolane from acyclic precursors. This bond construction choice enabled the use of chloroacetic acid, vinyl acetate, sodium thiosulfate and water to produce the oxathiolane. This simple process affords the hydroxyoxathiolane intermediate that can be converted into either lamivudine (3TC) or emtricitabine (FTC).
Supply Centered Synthesis to Current Late-State Intermediate Report